Egg diagnosis (PGT-M/PGT-SR)
PGT-M (Pre-implantation Genetic Testing for Monogenic/Single Gene Defects)/PGT-SR (Pre-implantation Genetic Testing for Chromosomal Structural Rearrangements) is a relevant option for a couple if the woman and/or the man has a known serious genetic disease, or a known risk of having a baby with a serious genetic disease or chromosome changes. The purpose of PGT-M/PGT-SR is to carry of the genetic diagnosis before transferring the embryo, to reduce the risk of the baby having a serious genetic disease or chromosome changes.
WE RECOMMEND PGT-M/PGT-SR IF:
- You and/or your partner is a carrier of or has a serious genetic disease or chromosome changes
- You’ve previously had to terminate a pregnancy due to serious genetic disease or chromosome changes in the foetus
- You’ve given birth to a child with a serious genetic disease or chromosome changes
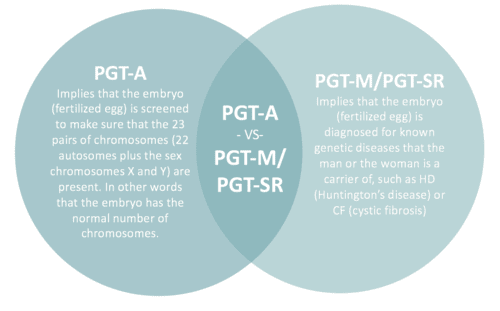
What's the difference between PGT-M/PGT-SR and PGT-A?
How is PGT-M/PGT-SR performed?
After egg retrieval and fertilization, your eggs will be cultivated in the lab for 5-6 days, to the blastocyst stage. Then each egg (blastocyst) is biopsied by taking out about 5 cells from the part of the egg that will later develop into the placenta. Blastocyst cultivation is necessary to perform the biopsy because it’s not until this stage that it’s possible to tell apart the inner cell mass (which develops into the foetus) and the outer cell mass (which develops into the placenta).
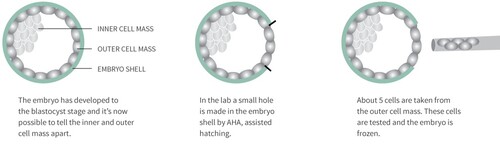
Once the biopsy has been performed, the embryo is frozen, and the cells taken from the outer cell mass are tested for the genetic disease or chromosome changes that you or your partner is a carrier of, before the embryo is transferred for implantation. Hence the name pre-implantation genetic diagnosis, PGT-M/PGT-SR.
The testing is carried out with the help of CooperGenomics, England, a lab specializing in genetics diagnosis. They help us identify and reject any embryos that would have developed into a diseased foetus, which means we only transfer embryos that have been proved to be free of the disease in question.
Will my embryos be harmed by freezing and thawing?
Blastocysts are very stable when it comes to freezing and hardly ever perish due to freezing and thawing. The chance of pregnancy when transferring thawed blastocysts is almost as high as when using fresh blastocysts (see our results here).
Preconditions for using PGT-M/PGT-SR
ICSI
PGT-M/PGT-SR can only be used in combination with micro-insemination or ICSI (Intracytoplasmic sperm injection) (ICSI). The difference between ordinary IVF treatment and ICSI lies in the way the egg is fertilized. In ordinary IVF, egg and sperm are simply mixed and it’s up to the sperm cells to find their way to the egg. In ICSI a single sperm cell is injected directly into the egg. This is the only way to be sure that the biopsy of the embryo contains genetic material from no other sperm cells than the one that fertilized the egg.
More eggs than in ordinary IVF
In ordinary IVF treatment, 8-9 eggs are retrieved on average. However, when planning for treatment using PGT-M/PGT-SR it’s a good idea to retrieve a few extra eggs. There are two reasons for this. Firstly, there is the fact that about 5% of chromosome test results tend to be inconclusive. And secondly, there will be a need to reject more eggs, exactly because some of them are carriers of the genetic disease or chromosome changes tested for.
Is PGT-M/PGT-SR a new method?
The PGT-M/PGT-SR method has been used in Denmark for almost 20 years. Besides Aagaard Fertilitetsklinik, treatment using PGT-M/PGT-SR is offered at Aalborg Universitetshospital and Rigshospitalet. Johnny Hindkjær, head of the laboratory at Aagaard Fertilitetsklinik, was involved in the development and approval procedure of the method in Denmark, and thus has extensive experience with PGT-M/PGT-SR treatment. Johnny holds degrees in biology and chemistry, and before joining Aagaard Fertilitetsklinik he worked as head of the laboratory at the fertility clinic of Aarhus Universitetshospital. The members of our PGT-M/PGT-SR and PGT-A team are – besides Johnny Hindkjær – Jørn Aagaard, our medical director, Malene Høeg, clinical embryologist, and Jens Michael Hertz, professor and senior hospital physician.
How safe is the method?
As we have a lot experience with the PGT-M/PGT-SR method, it’s a very safe treatment. The risk that the foetus, despite PGT-M/PGT-SR, turns out to be a carrier of the disease in question is as low as 1%. However, just to be even safer, we still recommend that you have a placental biopsy in week 11.
